QUIZ #1
Remember, you only need to know the 1st 20!
|
|
|
|
QUIZ #2
|
|
|
|
QUIZ 3 / THE "FINAL"
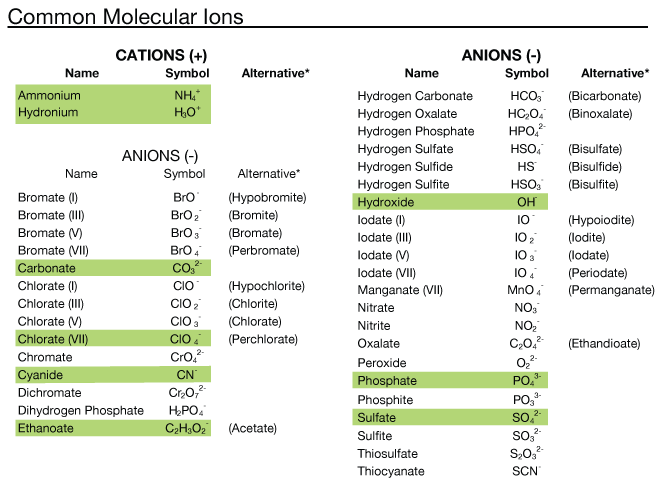
Common Molecular Ions
You can skip the Bromates and Iodates. Everything else is fair game for our last quiz. AND the green ones (except Hydronium because I didn't want to blow your mind) should already be memorized by now (post Christmas).
Note: Cation = positive charge Anion = negative charge
Note: Cation = positive charge Anion = negative charge
Practice Answer Key
|
| ||||||||||||
Click here to go to our QUIZLET page.
LAB - Either Ore
Redox? What does that mean?
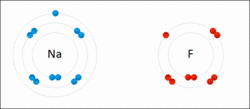
Sodium and fluorine bonding ionically to form sodium fluoride. Sodium loses its outer electron to give it a stable electron configuration, and this electron enters the fluorine atom exothermically. The oppositely charged ions are then attracted to each other. The sodium is oxidized, and the fluorine is reduced.
If the atom LOOSES electrons, it is oxidized.
If the atom GAINS electrons, it is reduced (the charge becomes more negative).
If the atom GAINS electrons, it is reduced (the charge becomes more negative).
LAB - What the Fizz
|
Before we go into lab, please take the time to work on the pre-lab:
Also, please visit this website to get familiar with what we are working on:
Fizzies Drink Tablets Family Page Here is the lab:
| |||||||||||||
And here is a sad clown.
LAB - A Latin Pile
| a_latin_pile_stdt_guide.pdf | |
| File Size: | 22 kb |
| File Type: | |
LAB - Our next project
|
We are making this...
|
Not this...
|
And definitely NOT THIS...
|
LAB - An edible cannon

From left to right: action of five Mentos candies (per bottle) with Perrier, classic Coke, Sprite and Diet Coke.
| mentos_explained.docx | |
| File Size: | 25 kb |
| File Type: | docx |
|
The easy way to engineer this.
|
And something for you to work on this summer.
|
Below this is older stuff. I have heard how the website is difficult to navigate and I am trying to reorganize to make it easier for you to find info.
Practice Problems
|
The Elements
I have set up a QUIZLET classroom to store all the vocab we will need to know as we go through the year. Your primary focus for now should be the first 20 elements in the periodic table as we will be dealing with them the most this year.
| ||||||||||||||||||||||||||||
Covalent & Ionic Bonds
Lab 1 - Acids, Bases, & pH: You can stop at 4 minutes. After that it's too much math!
|
Lab 2 - Chemical Equations
|
|
|
|
Types of Reactions:
Combination
Combination reactions, also known as synthesis reactions, involve the combination of two or more substances into a single new substance:
A + B ---> AB OR A + B ---> C
Many metal oxides, for example, absorb carbon dioxide (CO2) from the air to form carbonates:
CaO(s) + CO2(g) ---> CaCO3(s)
Decomposition
Decomposition reactions represent the reverse of the combination process in that one substance breaks down into two or more substances:
AB ---> A + B OR C---> A + B
Water, or H2O, for example, decomposes into its constituent elements when subjected to an electric current:
2 H2O(l) ---> 2 H2(g) + O2(g)
Combustion
Combustion reactions occur when substances, particularly those containing carbon, burn in the presence of oxygen to produce carbon dioxide and water:
CxHy + O2 ---> CO2(g) + H2O(g)
Methane, for example, with chemical formula CH4 combusts as:
CH4(g) + O2(g) ---> CO2(g) + 2 H2O(g)
Single-Replacement
Single-replacement reactions occur when an element replaces one of the elements in a compound:
A + BX ---> AX + B where X represents the atom being exchanged.
Many metals, for example, react with acids to produce hydrogen and a metal salt:
Zn(s) + 2 HCl(aq) ---> ZnCl2(aq) + H2(g)
Double-Replacement
Double-replacement reactions involve two compounds exchanging elements:
AB + CD ---> AD + CB
Double-replacement reactions commonly occur when one of the products is not soluble and precipitates out of the reaction solution as a solid:
CaCl2(aq) + Na2CO3(aq) ---> CaCO3(s) + 2 NaCl(aq)
Combination
Combination reactions, also known as synthesis reactions, involve the combination of two or more substances into a single new substance:
A + B ---> AB OR A + B ---> C
Many metal oxides, for example, absorb carbon dioxide (CO2) from the air to form carbonates:
CaO(s) + CO2(g) ---> CaCO3(s)
Decomposition
Decomposition reactions represent the reverse of the combination process in that one substance breaks down into two or more substances:
AB ---> A + B OR C---> A + B
Water, or H2O, for example, decomposes into its constituent elements when subjected to an electric current:
2 H2O(l) ---> 2 H2(g) + O2(g)
Combustion
Combustion reactions occur when substances, particularly those containing carbon, burn in the presence of oxygen to produce carbon dioxide and water:
CxHy + O2 ---> CO2(g) + H2O(g)
Methane, for example, with chemical formula CH4 combusts as:
CH4(g) + O2(g) ---> CO2(g) + 2 H2O(g)
Single-Replacement
Single-replacement reactions occur when an element replaces one of the elements in a compound:
A + BX ---> AX + B where X represents the atom being exchanged.
Many metals, for example, react with acids to produce hydrogen and a metal salt:
Zn(s) + 2 HCl(aq) ---> ZnCl2(aq) + H2(g)
Double-Replacement
Double-replacement reactions involve two compounds exchanging elements:
AB + CD ---> AD + CB
Double-replacement reactions commonly occur when one of the products is not soluble and precipitates out of the reaction solution as a solid:
CaCl2(aq) + Na2CO3(aq) ---> CaCO3(s) + 2 NaCl(aq)
Lab 3 - Chromatography
|
|
|
|
Lab 4 - Copper into Gold
|
Questions to help you with your thinking...
What determines the type of atom? In other words, which part of the atom determines what kind of element it is?
How could we physically/chemically test our coins at each step?
The reaction on the right is not the same one that we did, but it is similar. Could you use what happens in that video to explain what is happening in our lab?
|
|