Week 25: 2/20 - 2/24
|
| ||||||||||||
|
|
|
|
|
|
Week 20: 1/16 - 1/20
|
| ||||||
|
|
|
Week 19: 1/9 - 1/13
|
|
|
|
|
|
Week 15: 12/12 - 12/16
| quiz_prep_calorimetry_6___7_complete_w_answer_key.pdf | |
| File Size: | 193 kb |
| File Type: | |
|
|
|
|
|
|
Week 13: 11/28 - 12/2
|
|
|
|
|
If you have the time, watch this. He's way better than I am!
Week 11: 11/14 - 11/18
|
| ||||||
Week 10: 11/7 - 11/11
Week 9: 10/31 - 11/4
HAPPY HALLOWEEN!
Since we will not have class on Monday, please work on these over the extended weekend.
Do exercises 4.45, 4.47, 4.49, 4.51, & 4.53 There are two “stretch" exercises that I’d recommend as they will get you thinking about the “activity series” I talked about in class: 4.55, 4.57
Do your best, talk to each other, get in study groups of 3-6 people, and do your best.
The homework should be stress free in that you aren't going to be graded on it if you make a mistake.
Email with questions. (Note: I will be inverting the order of topics on the website so that the newest stuff is on top.)
Since we will not have class on Monday, please work on these over the extended weekend.
Do exercises 4.45, 4.47, 4.49, 4.51, & 4.53 There are two “stretch" exercises that I’d recommend as they will get you thinking about the “activity series” I talked about in class: 4.55, 4.57
Do your best, talk to each other, get in study groups of 3-6 people, and do your best.
The homework should be stress free in that you aren't going to be graded on it if you make a mistake.
Email with questions. (Note: I will be inverting the order of topics on the website so that the newest stuff is on top.)
|
11/1
If you didn't write the homework down in class, the problems for tonight are: 4.61, 63, 65, 69, 71 I feel like I missed one or two, and I forgot to write them down before I left for the day, so you lucked out. |
| ||||||
Week 1: 9/6-9/9
|
Week 2: 9/12-9/16
| ||||||||||||||||||||||
This guy is British, and some of the graphics or music is hokey, but it is solid. Especially if you need another/different explanation.
|
|
|
|
Week 3: 9/19 - 9/23
|
|
| ||||||||||||
|
|
|
|
|
|
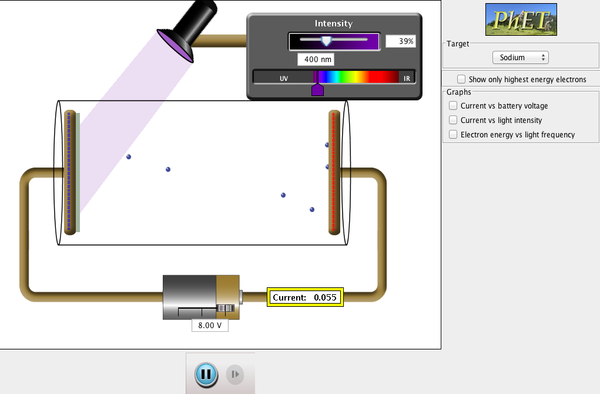
Photoelectric Effect
One of the last questions in the homework asks you to explain Einstein's reasoning for the photoelectric effect (i.e. Why was violet light able to force electrons to move but red light was not?)
Red light lacks enough energy to "dislodge" an electron.
|
Visible light is approximately:
|
Other EM is approximately:
|
courtesy of: http://physics.info/photoelectric/
In 1905, Einstein realized that light was behaving as if it was composed of tiny particles (initially called quanta and later called photons) and that the energy of each particle was proportional to the frequency of the electromagnetic radiation that it was a part of. What we perceive as a continuous wave of electromagnetic radiation is in reality a stream of discrete particles.
Before Einstein's explanation, the photoelectric effect was a real mystery. Scientists couldn't really understand why low-frequency high-intensity light would not cause electrons to be emitted, while higher-frequency low-intensity light would. Knowing that light is made up of photons, it's easy to explain now. It's not the total amount of energy (i.e., the intensity) that's important, but the energy per photon.
In 1905, Einstein realized that light was behaving as if it was composed of tiny particles (initially called quanta and later called photons) and that the energy of each particle was proportional to the frequency of the electromagnetic radiation that it was a part of. What we perceive as a continuous wave of electromagnetic radiation is in reality a stream of discrete particles.
Before Einstein's explanation, the photoelectric effect was a real mystery. Scientists couldn't really understand why low-frequency high-intensity light would not cause electrons to be emitted, while higher-frequency low-intensity light would. Knowing that light is made up of photons, it's easy to explain now. It's not the total amount of energy (i.e., the intensity) that's important, but the energy per photon.
Week 4: 9/26 - 9/30
|
| ||||||||||||
Week 5: 10/03 - 10/07
EXTRA ONLINE PRACTICE - Nomen.
|
| ||||||||||||||||||
Week 6: 10/10 - 10/14
|
EXTRA ONLINE PRACTICE - Balancing Equ.
|
Note: I'm moving the test to Week 7, we still need to cover Acid/Base this week and Redox reactions.
AND I'm moving the quiz on ionic equations (10/12) back to Thursday (10/13).
AND I'm moving the quiz on ionic equations (10/12) back to Thursday (10/13).
|
It's a little tricky (tricky, tricky, tricky!). The best thing you can do is look at the problem and write down everything your know.
| ||||||||||||||||||||||||||||||||
|
|
|
Week 7: 10/17-10/21 Redox! and our 1st test!!!
|
|
|
More online resources!
Remember! Part of any AP class is learning how to direct your own learning. Do you: read the textbook, watch videos, take notes (either out of the book, or in class), get together with your peers in study groups, come ask for more help?
|
Week 8: 10/24 - 10/28 Redox in Acidic & Basic Solutions
|
| ||||||||||||
|
|
|
An easy way to predict what reaction will take place when two chemicals are mixed is to identify the type of reaction that's likely to occur when the chemicals are combined. We were going to cover this topic later this week, but why wait. Right?!?
NOTE: The last one is a special case of double replacement, if you will.
1) Combustion: [some compound with C and H] + O?? CO? + H?O. This reaction is exothermic and the water is in the form of water vapor.
2) Synthesis: A + B --> C. Put another way, one compound is made from several others.
3) Decomposition: C --> A + B. Put another way, one compound breaks apart to form several others.
4) Single displacement: A + BC --> B + AC. A pure element switches places with an element in a chemical compound. This is usually (but not always) a metal.
5.a) Double displacement: AB + CD --> AD + CB. The cations on two ionic compounds switch places.
5.b) Acid-base: HA + BOH --> BA + H?O. This is a double displacement reaction that makes water.
*If two ionic compounds are combined, it's usually safe to predict that a double displacement reaction will occur.
*If the chemicals mixed are oxygen and something containing carbon, it's usually a combustion reaction.
*If we start with only one reactant, the reaction taking place is probably a decomposition reaction. To predict the products of such a reaction, see what happens if the chemical breaks into smaller, familiar products such as water, carbon dioxide, or any of the gaseous elements.
*When pure elements are combined, synthesis reactions are the frequent result.
*If a pure element combines with an ionic compound, a single displacement reaction may take place.
*If a compound containing the hydroxide ion is involved, check the other compound to see if it contains hydrogen. If it does, it may be an acid-base reaction.
(We haven't talked about this yet, but we will eventually.)
*In order for a single displacement reaction to occur, the pure metal has to be higher on the activity series than the metal already in a compound.
K > Na > Li > Ba > Ca > Mg > Al > Zn > Cr > Fe > Ni > Sn > Pb > Cu > Ag
Most Active Least Active
(Last Tip!)
In order for a double displacement reaction to occur, the following conditions must be met:
1) Both of the reagents must be soluble in water: If you can?t dissolve the reagents, they won?t be able to react with each other.
2) One (and only one) of the products must be insoluble (a solid) in water. If both are solids, there is a reaction but it consists of a bunch of gunk all mixed together, giving us unusable product. If both are aqueous, then you just have a big ionic soup with no real compound at all. For the reaction to be successful, one compound must be easily separated from the other, as is the case when one has dissolved and the other has not.
NOTE: The last one is a special case of double replacement, if you will.
1) Combustion: [some compound with C and H] + O?? CO? + H?O. This reaction is exothermic and the water is in the form of water vapor.
2) Synthesis: A + B --> C. Put another way, one compound is made from several others.
3) Decomposition: C --> A + B. Put another way, one compound breaks apart to form several others.
4) Single displacement: A + BC --> B + AC. A pure element switches places with an element in a chemical compound. This is usually (but not always) a metal.
5.a) Double displacement: AB + CD --> AD + CB. The cations on two ionic compounds switch places.
5.b) Acid-base: HA + BOH --> BA + H?O. This is a double displacement reaction that makes water.
*If two ionic compounds are combined, it's usually safe to predict that a double displacement reaction will occur.
*If the chemicals mixed are oxygen and something containing carbon, it's usually a combustion reaction.
*If we start with only one reactant, the reaction taking place is probably a decomposition reaction. To predict the products of such a reaction, see what happens if the chemical breaks into smaller, familiar products such as water, carbon dioxide, or any of the gaseous elements.
*When pure elements are combined, synthesis reactions are the frequent result.
*If a pure element combines with an ionic compound, a single displacement reaction may take place.
*If a compound containing the hydroxide ion is involved, check the other compound to see if it contains hydrogen. If it does, it may be an acid-base reaction.
(We haven't talked about this yet, but we will eventually.)
*In order for a single displacement reaction to occur, the pure metal has to be higher on the activity series than the metal already in a compound.
K > Na > Li > Ba > Ca > Mg > Al > Zn > Cr > Fe > Ni > Sn > Pb > Cu > Ag
Most Active Least Active
(Last Tip!)
In order for a double displacement reaction to occur, the following conditions must be met:
1) Both of the reagents must be soluble in water: If you can?t dissolve the reagents, they won?t be able to react with each other.
2) One (and only one) of the products must be insoluble (a solid) in water. If both are solids, there is a reaction but it consists of a bunch of gunk all mixed together, giving us unusable product. If both are aqueous, then you just have a big ionic soup with no real compound at all. For the reaction to be successful, one compound must be easily separated from the other, as is the case when one has dissolved and the other has not.