Labs
As we get up and running, I will begin to add pertinent (relative, topical) videos and pre-labs to this page.
Lab Reports
|
The whole point of this class is to learn how to write lab reports. We will not do this right away. First we will learn how to keep a lab notebook, then we will learn to write a lab report. You can see the lab report guide here. (On the right.)
|
| ||||||
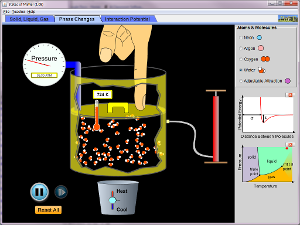
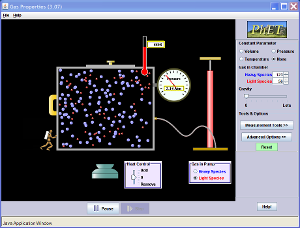
Activity 1 Solid, Liquid, or Gas
Hello all,
This is our first week together and I don't know very much about you or your skill level.
Today's "lab" will be the easiest one we do all semester, and it will tell me very much about you, your skill level, how well you write, and what I need to do to help you learn.
If you have time, please visit this website in order to get ready for class.
Or you can play with the simulation below.
This is our first week together and I don't know very much about you or your skill level.
Today's "lab" will be the easiest one we do all semester, and it will tell me very much about you, your skill level, how well you write, and what I need to do to help you learn.
If you have time, please visit this website in order to get ready for class.
Or you can play with the simulation below.
|
Here is a copy of week 1's lab if you loose it or need another one.
If you can't get the simulation to run, you can go to their website HERE and download the simulation and run it on your computer.
| |||||||
Week 2
Since we did not get very far into lab, week 2 will continue the 1st lab.
Week 3
|
For my 8th and 9th graders:
Please download this exercise and answer the questions to the best of your ability. If you save it as a word file on your computer, you can submit it digitally via/through this website and avoid printing it! |
| ||||||
Week 4
|
Blowing stuff up.
Your question for this week, "How do you make/generate/get Hydrogen gas?" Some resources
|
|
|
If you cannot get the simulation on the left to run, you have two options:
1. Download the file HERE and run it on your computer.
2. Go to the University of Colorado's website HERE and click on the "RUN NOW" button to play it off of their website.
| |||||||
Week 5
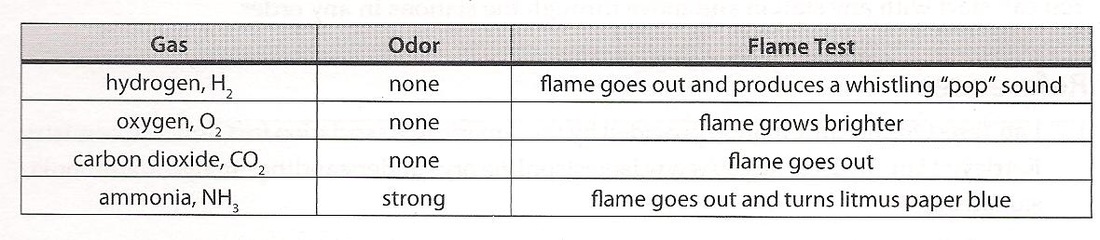
What gas did we make?
Week 6
|
We will have our Element Quiz this week.
|
Go HERE for the on-line practice.
|
Week 7
We will complete our lab write-up on all the chemicals we tested for pH.
Week 8
|
We will test our unknown this week.
Don't get scared by what you are about to read, you know some of this and the rest we will get to over the course of the year! When we test our unknown, we will do one of these five reactions: Combination Combination reactions, also known as synthesis reactions, involve the combination of two or more substances into a single new substance: A + B ---> AB Many metal oxides, for example, absorb carbon dioxide (CO2) from the air to form carbonates: CaO(s) + CO2(g) ---> CaCO3(s) Decomposition Decomposition reactions represent the reverse of the combination process in that one substance breaks down into two or more substances: AB ---> A + B Water, or H2O, for example, decomposes into its constituent elements when subjected to an electric current: 2 H2O(l) ---> 2 H2(g) + O2(g) Combustion Combustion reactions occur when substances, particularly those containing carbon, burn in the presence of oxygen to produce carbon dioxide and water: CxHy + O2 ---> CO2(g) + H2O(g) Methane, for example, with chemical formula CH4 combusts as: CH4(g) + O2(g) ---> CO2(g) + 2 H2O(g) |
Single-Replacement
Single-replacement reactions occur when an element replaces one of the elements in a compound: A + BX ---> AX + B where X represents the atom being exchanged. Many metals, for example, react with acids to produce hydrogen and a metal salt: Zn(s) + 2 HCl(aq) ---> ZnCl2(aq) + H2(g) Double-Replacement Double-replacement reactions involve two compounds exchanging elements: AB + CD ---> AD + CB Double-replacement reactions commonly occur when one of the products is not soluble and precipitates out of the reaction solution as a solid: CaCl2(aq) + Na2CO3(aq) ---> CaCO3(s) + 2 NaCl(aq) |
Week 9 - Exam Prep
|
Test Practice
Hi, On the right hand side you will see two files. The top file has practice problems so you can get more experience balancing equations. The bottom file is the answer key. I strongly urge you to tr the problems, ask your parents for help, or email me for help before you look at the answer key. After you have done the problems to the best of your ability, look at the answer key. If you still don't understand what you did wrong, email me. I will check my email every day until next Wednesday. |
| ||||||||||||||||||